A walk through the timeline of 2,3,4-Trihydroxybenzophenone shows how chemistry finds possibilities everywhere. People started making and studying benzophenones a century ago, trying to pull out new colors and medicinal properties from those aromatic rings. By the time researchers figured out how to attach hydroxy groups to different spots on the structure, a new world opened up. 2,3,4-Trihydroxybenzophenone didn't pop up at random—curiosity drove scientists to ask what would happen if three hydroxyls came together on a benzophenone backbone. The chemical literature grew steady, feeding off discoveries around polyphenols and antioxidants through the 20th century. Today, anyone who looks into natural product chemistry or advanced organic synthesis will run into derivatives like this one, which keep showing up in surprising biological and material applications.
Looking at 2,3,4-Trihydroxybenzophenone, what stands out is its flexibility as a building block. Its structure—two phenyl rings connected by a carbonyl, with three hydroxyl groups packed tightly on one ring—makes it a favorite for folks in medicinal and materials science alike. Chemists keep reaching for it in the lab, chasing new antioxidants, UV absorbers, and smart polymers. It shows up in both natural and synthetic research, which tells me there's more to learn about how it might shield cells from damage or make plastics tougher. When someone mentions work on hydroxybenzophenones, it's clear this isn’t just another catalog chemical—it's a point of entry into tough scientific questions.
In the lab, you can spot 2,3,4-Trihydroxybenzophenone from its off-white to tan solid appearance and modest water solubility, a trait tied to its aromatic rings and multiple hydroxy groups. You’ll find it dissolving best in alcohols or acetone—environments where hydrogen bonds can really go to work. The hydroxyls wonder around the carbon backbone, donating electrons and making the structure less stable in basic conditions. I've watched colleagues run melting point experiments, usually finding it decomposes rather than producing a steady liquid. Its rich aromatic core loves UV light, which pushes chemists to test its potential in sunscreen chemistry and as a protective pigment.
2,3,4-Trihydroxybenzophenone rarely comes with complicated packaging. Labels tend to cite its CAS number and molecular formula, with purity levels ranging from low nineties to almost analytical standards. Labs and suppliers usually work with crystalline powders, and those who handle it need to check for finely divided particles drifting through the air. Chemists reading the label see hints at storage temperature and advisories about sealing to guard against excess humidity, as moisture can slowly degrade sensitive polyphenolic compounds. High-performance liquid chromatography keeps the quality in check, and infrared spectra, mass spec, and NMR data are used to confirm that no one is selling the wrong isomer or, worse, a by-product.
Synthesis of 2,3,4-Trihydroxybenzophenone isn’t the hardest, but it pays to get the details right. People often start with a properly substituted benzoyl chloride and react it with a hydroxybenzene under Friedel-Crafts acylation conditions, often with an aluminum chloride catalyst. From there, clever hydrolysis and protection group strategies can leave the hydroxy groups in the right spots. More recently, some labs have moved to milder routes, turning to oxidative coupling or directed ortho-metalation, cutting waste and giving better yields. It’s easy to burn time on purification if your side-products include mono- or dihydroxy analogs, so experience with column chromatography comes in handy. As someone who's tried wrangling polyhydroxy aromatics, cleaning up these products—getting rid of colored impurities and trace metals—takes as much care as the synthesis itself.
If a molecule ever invited modification, it’s this one. The adjacent hydroxy groups offer sites for methylation, esterification, or plain old oxidation. Chemists walk a fine line between tuning solubility and locking down the triple-hydroxy setup that gives the molecule its unique properties. Turning it into ethers or esters changes everything from solubility to biological activity, and it’s not uncommon to see research spinning off into new benzophenonic acids and polymers through further reactions. In biochemistry, I’ve seen work where its phenolic sites undergo enzymatic transformations, hinting at mild catalytic cycles that produce rare metabolites. Coordination chemistry plays a role here, too; the oxygens on this molecule aren’t shy, and they bind metals tightly, making for interesting complexes that pop up in catalysis and environmental remediation labs.
In research circles, clear naming means less confusion. Besides its full name, you’ll often bump into "Benzophenone-1," "2,3,4-Trhydroxybenzoylbenzene," and sometimes the shorthand "THBP." Each name tells you where the hydroxy groups sit, ensuring no one mistakes it for a cousin with substitutions in different spots. I’ve seen suppliers list alternate IUPAC labels, and sometimes older literature swaps in less systematic names, so a chemist quickly learns to check structures, not just names, when comparing studies or ordering stock.
Dealing with this chemical out of the literature throws up less risk than, say, strong acids or volatile organics, but it's not wise to treat it like sugar. Fine particulate dust can irritate the lungs if inhaled, especially in tight labs without proper ventilation. Its phenolic hydroxyl groups suggest a weak acidic profile, which in my experience, means you should avoid unnecessary skin contact to sidestep possible sensitization. Proper PPE—gloves, goggles, lab coat—keeps exposure minimal. Safety data gets updated periodically, and people working with the compound take protocols seriously, especially when scaling jobs or experimenting with novel derivatives that haven’t been exhaustively tested for chronic effects. Waste disposal follows standard organic rules, collecting residues for designated chemical waste streams.
If research tells us anything, it’s that 2,3,4-Trihydroxybenzophenone isn’t defined by just one use. Its antioxidant activity attracts teams working on skincare and anti-aging products, who see polyphenols as potential shields against UV-driven cell damage. Analytical chemists test it in methods for detecting transition metals, where its chelation skills come in handy. Synthetic organic chemists appreciate its function as a precursor for more complex ligands, monomers in advanced materials, and specialty sensors. In my work, I've seen it pop up in exploratory trials for drug discovery, usually as a lead compound for enzyme inhibition studies and sometimes in antimicrobial projects that target resistant pathogens. Within material sciences, researchers value the molecule’s UV-absorbing capabilities, trying to plug it into coatings and plastics destined for outdoor use, extending product longevity where sunlight tries to break things down.
Curiosity drives most R&D around this benzophenone. Academic groups still run bench experiments on antioxidant capacity, striving to link structure with cellular activity using tests like DPPH scavenging and ABTS assays. Some teams shift toward finding environmentally benign synthesis routes, reducing solvent use and ditching heavy metal catalysts, again proving that green chemistry creates its own wave of innovation. In drug research labs, the focus shifts to analog development—adding or swapping functional groups to finesse bioactivity. Material scientists are turning out polybenzophenone polymers, hoping for breakthrough applications in electronics or smart packaging. The data keeps growing, but the challenge remains the same: how to move from academic promise to scalable, profitable application.
Scientists make sure they know a molecule’s risks, and for 2,3,4-Trihydroxybenzophenone, the picture isn't all clear yet. Most studies echo general polyphenol safety data, suggesting acute toxicity is low in cell culture or animal models at reasonable doses, but the story shifts as exposure mounts. Chronic toxicity, long-term environmental fate, and bioaccumulation all draw questions that researchers haven’t finished answering. Some benzophenones have caught flak for endocrine-disrupting effects, and while trisubstituted versions look less potent, nobody skips caution, particularly with products designed for daily human contact. Environmental chemists watch for breakdown products leaching into soil and water, triggering further rounds of evaluation.
Looking ahead, 2,3,4-Trihydroxybenzophenone isn’t running out of uses or mysteries. As research digs deeper into polyphenolic therapeutics, designers keep shuffling molecules like this one through screening pipelines, chasing more effective drugs and cosmetics. With sustainable chemistry in the spotlight, future methods could move toward biocatalysis or continuous flow synthesis, trimming waste and boosting yields. In the electronics field, there's real hope for tailored polymers based on this skeleton, promising performance and durability no one could wring from older materials. I expect to see more environmental monitoring, as researchers figure out not just how to use it, but how to clean up when it’s used in bulk. The push for transparency in safety testing and labeling will keep suppliers on their toes. As far as small molecules go, 2,3,4-Trihydroxybenzophenone won’t be fading quietly into a database; it’s set to have a say in how we make, use, and regulate specialty chemicals in the coming years.
People outside of chemistry circles might not catch the buzz around 2,3,4-Trihydroxybenzophenone, but for those who spend days at the bench or walk factory floors, this molecule holds a hidden weight. In plain terms, this compound steps into the shoes of a versatile chemical assistant. It pulls its weight in the science and manufacturing worlds for more than just its structure or fancy name.
Researchers dig into 2,3,4-Trihydroxybenzophenone mostly for its talents as an intermediate. The benzophenone backbone, paired with three hydroxyl groups, opens doors in organic synthesis, especially where unique fragrances, pharmaceutical precursors, or UV-absorbing polymers demand precision. I’ve seen chemists reach for it when building complex molecules that eventually touch the lives of people on grocery store shelves, hospital beds, or even inside car dashboards.
One direct application comes up in sunscreen development. Structures related to 2,3,4-Trihydroxybenzophenone have a knack for absorbing ultraviolet light. While this specific molecule might not go into popular lotions, similar ones help block harmful rays. Researchers will sometimes tweak this compound in the lab to chase better UV protection or pursue allergy-friendly solutions, as the search for safer and more effective sunblock never really ends.
Not every chemical gets singled out in peer-reviewed papers. 2,3,4-Trihydroxybenzophenone sometimes helps scientists mimic the natural world, especially the activities of plant phenolic compounds, which play defensive roles in leaves and stems. This connection gives researchers a handle on plant biochemistry or ways to tap into antioxidant properties for future applications in foods or nutraceuticals. I’ve watched food scientists explore these directions, hoping to bolster shelf life or add a health twist without loading up on artificial preservatives.
The importance of 2,3,4-Trihydroxybenzophenone does hit a snag if handled without care. Like many fine chemicals, this substance calls for respect and safe handling. Environmental policy and worker safety are front-and-center topics in production labs. Stories of mishaps echo reminders that stringent oversight isn’t just a bureaucratic headache—it saves livelihoods and ecosystems. If manufacturing ramps up, proper waste management and real transparency about supply chain risks make a difference.
Looking ahead, research holds the key to responsible use and more effective impact from compounds like 2,3,4-Trihydroxybenzophenone. Collaborative science between universities, industry, and regulators can sniff out better synthesis routes, greener solvents, and scalable methods that avoid the pitfalls of hazardous byproducts. Chemical engineers and environmental scientists who step beyond their silos can shape safer compounds that keep up with the rapid demands of technology, healthcare, and consumer goods.
The story of 2,3,4-Trihydroxybenzophenone might not hit the evening news, but it drifts into medicine bottles, plastic casings, and food packages more often than most people realize. As the world leans further into high-performance materials and health-conscious living, this quiet workhorse reminds us why detailed knowledge and ethical stewardship matter in every stage from lab bench to marketplace.
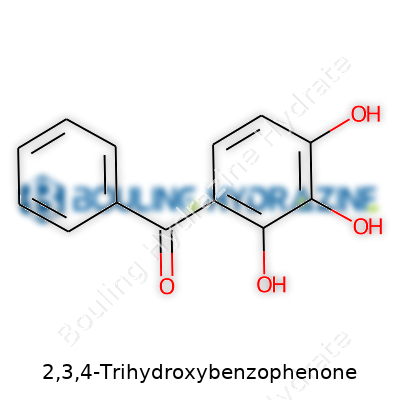
The name 2,3,4-Trihydroxybenzophenone looks like something out of a chemistry textbook, but beneath the jargon, lies a molecule with a simple heart: it’s a benzophenone with three hydroxy groups tacked to one of the benzene rings. If you picture the benzophenone skeleton, you’re looking at two benzene rings joined by a ketone group. With this compound, the numbers 2, 3, and 4 mean you find those OH (hydroxy) groups lined up next to each other on one of those rings. This arrangement makes the molecule quite active compared to its lazier cousins that don’t carry so many hydroxy tags.
Years spent in research labs taught me that the shape and decoration of a molecule decide everything about what it can do. With three hydroxy groups side-by-side, 2,3,4-Trihydroxybenzophenone can form a network of hydrogen bonds both within itself and with other compounds. Picture velcro pads ready to stick to anything with a charge or a polar side. This potential for tight bonding gives the compound its antioxidant bite. I’ve seen its cousins, like oxybenzone, hit the sunscreen world because of similar traits—blocking out harmful UV rays by gobbling up their energy and spitting it back out as harmless heat.
By now, chemists know that basic benzophenone gets a lot of attention as a building block in various fields, but adding more hydroxy groups can ramp up biological activity. That activity comes with some trade-offs. On one hand, molecules like 2,3,4-Trihydroxybenzophenone can serve as powerful antioxidants in food science and cosmetics. They help slow down oxidative reactions that mess up fresh food color or cause oils to go rancid. Some researchers have looked into this structure for its possible role in natural plant defense mechanisms, too, since so many phenolic compounds show up in nature’s arsenal against pests and diseases.
Despite its active structure, molecules like 2,3,4-Trihydroxybenzophenone don’t land in products as often as they could. I’ve seen two big reasons for this hesitation. For starters, adding more OH groups makes the molecule less likely to dissolve in oils. Many industrial processes and cosmetic formulations count on good oil solubility, so formulators often pick structures that blend in better with their base materials. The next catch is that a structure built for high reactivity (like this one with three hydroxy groups) can sometimes cause skin irritation or break down into products that give regulatory bodies pause. These concerns don’t mean the molecule is trouble by default, but they do raise standards for toxicological testing and purity controls.
What’s the workaround? For scientists in academia and industry, building up safety data through careful studies wins trust. Supplementing synthetic routes with green chemistry can also minimize side products and waste. And by combining this molecule in smart ways—either sticking to the right concentration or blending it with gentler materials—there’s a shot at unlocking its power without stirring up worries. Every time we push for a balance between effectiveness and safety, compounds like 2,3,4-Trihydroxybenzophenone keep teaching that structure isn’t just about lines on paper. It shapes what’s possible, what’s wise, and what might come next in chemical innovation.
Chemistry rarely delivers simple answers, especially when it comes to handling compounds like 2,3,4-trihydroxybenzophenone. People working in labs or the chemical industry don’t always get a say about what’s in front of them, but safety needs to come before convenience each time. With this particular compound, often found as a powder or crystalline solid, using good sense, basic training, and some practical rules matters more than marketing claims or ambiguous assurances.
2,3,4-trihydroxybenzophenone isn’t some explosive nightmare, but that doesn’t make it harmless. It’s easy to overlook risks if you handle small amounts or only see it as part of a research project, but irritations and mild toxicity reports shouldn’t get dismissed casually. My own early days in the university lab taught me the hard way that skipping gloves or skipping goggles isn’t just a rookie mistake—it’s an invitation for skin rashes and eye irritation. Any chemical with multiple phenolic groups tends to be irritating to skin and eyes, and there’s no magic bullet to change that.
Existing research lists several benzophenone derivatives as mild-to-moderate skin and respiratory irritants. There are well-documented reports of skin redness or even allergic responses from repeated or careless handling. The hydroxy groups add another layer—these compounds tend to be more soluble and interact more readily with tissues. If inhaled as a fine powder, particulates can irritate airways, especially for people with asthma or allergy histories. Accidental ingestion can also irritate the gut, though acute poisoning is rare for this particular compound. The available data from resources like the PubChem and European Chemicals Agency highlight these practical risks, not catastrophic emergencies.
None of the main occupational safety agencies, like OSHA or NIOSH in the United States, list 2,3,4-trihydroxybenzophenone as a high-hazard substance. That doesn’t mean you can toss caution out the window. Routine safety data sheets recommend gloves and eye protection on the logic that irritation is the most likely problem. Even if something isn’t regulated as a carcinogen or acute toxin, repeated exposure can lead to chronic symptoms or set off skin issues that stick around long after you stop the work.
Many accidents happen because someone gets comfortable or assumes the risk is trivial. I’ve seen experienced researchers take shortcuts with “low-toxicity” organics, only to spend the next week treating contact dermatitis. Best practice means using gloves, working under a fume hood, avoiding inhalation, and keeping food far away from the workbench. Spills need prompt clean-up, and any splash should send you straight to the eye-wash or safety shower.
Many health and safety campaigns push for training and regular reminders. That’s not just bureaucratic box-ticking. It helps keep good habits alive. If you’re new, listen to supervisors and read the material safety data sheets carefully. Lab managers can provide safer alternatives or extra personal protective equipment if there’s a clear need.
Handling 2,3,4-trihydroxybenzophenone isn’t about paranoia but about respecting learned lessons and the few documented risks. Exposure control, common sense, and taking nothing for granted keeps both novices and veterans protected. Claims of “no significant hazard” all too often ignore the real world, where carelessness turns minor irritants into memorable injuries.
Ask any chemist about storage, and you’ll usually hear some variation on “keep it dry and keep it cool.” That’s not just caution or habit; it’s hard-won wisdom. 2,3,4-Trihydroxybenzophenone, with its multiple hydroxyl groups, fits that advice pretty well. Moisture and heat play the troublemakers here. With a molecule like this, the hydroxyls want to grab onto water from the air. I’ve watched similar compounds clump up or start showing brown edges in bottles that looked like they’d been through a few bad summers. There’s almost a sense of betrayal—you return to a once pristine powder, only to find it’s a sticky mess.
Too much exposure to air, especially in a humid lab or stockroom, is just asking for slow but steady deterioration. Hydroxyls pull in water, sure, but oxygen can nudge things in the wrong direction too. Oxidative changes might not always be obvious at first glance, but purity drops before long. That means stuff like brownish streaks or odd smells in what should be a plain, off-white powder.
After years of working with this class of chemicals, I would never stash 2,3,4-Trihydroxybenzophenone near a window or strong light source. Some molecules break down under light, especially if there’s a trace of metals or peroxides nearby. Amber bottles or locking away in opaque bins helps keep the sun’s rays out, even if the lights in most labs barely flicker.
It’s hard to overstate how much difference a dry storage cabinet makes. I’ve lost good material to humid college basements, and every scientist remembers at least one lesson learned the tough way. If you have access to dedicated desiccators or cabinets with humidity control, use them. Silica gel packets might seem old-school, but they buy extra time when the atmosphere decides to turn muggy. Sealing containers tightly, even double-bagging, shows its worth almost immediately.
Room temperature usually works if things don’t swing wildly, but somewhere between 2°C and 8°C tends to be safer for longer-term storage. Once, after leaving a batch in a shared fridge, I discovered condensation thanks to careless opening and closing. So, label containers clearly and remind staff or students not to treat chemical fridges like the break room.
Most issues with 2,3,4-Trihydroxybenzophenone don’t begin with the molecule itself—they stem from small shortcuts or oversight. People often grab what they need, leave the bottle open a minute too long, or forget to check the condition of their desiccant. Over time, those choices steal reliability from your work. Analytical results get uncertain. Waste builds up. There’s no quick fix, only steady prevention.
It helps to keep an up-to-date inventory and toss out anything suspect. Paying a little more attention to tight lids and dry hands at the bench pays back in fewer headaches downstream. In the end, preserving chemicals like 2,3,4-Trihydroxybenzophenone isn’t glamorous or high-tech, just common-sense respect for the craft.
Most folks wouldn’t give much thought to a molecule weighing just a tick on the atomic scale, but in chemistry, those numbers change everything. With 2,3,4-Trihydroxybenzophenone, the molecular weight lands at 230.21 grams per mole. That number has the power to shape how researchers approach synthesis, dosing in research, and even safety protocols.
During my school years, calculating molecular weights felt like busywork, but the further you dig into chemistry labs, the clearer it gets: a misplaced decimal or skipped atom can break an experiment or lead to hazardous mistakes. Each element sits there on the periodic table, carrying a weight that science hashed out with a lot of precision—hydrogen at about 1.01, carbon at 12.01, oxygen at 16, and so on.
For anyone fresh to this, 2,3,4-Trihydroxybenzophenone packs a core of carbon and hydrogen, with extra oxygen on the ring. Structural formulas aren’t just pretty pictures; they let you tally up: 13 carbons (13 x 12.01), 10 hydrogens (10 x 1.01), and 4 oxygens (4 x 16.00). Adding those up brings you smack to 230.21. That’s not a suggestion or a rounded guess — it’s a quantity that affects how much to measure out, how to mix solutions, and the expectations with chemical reactions.
Beyond textbook exercises, people working in pharmaceuticals or research trust these molecular weights to build drugs safely. A miscalculated weight, even by one digit, sets off a chain of errors. Take drug formulation — it isn’t a matter of sprinkling powder into capsules. Doses depend on molecular weights. Too much of an active compound, toxicity threatens. Too little, and treatments fall flat. That’s not just science talk; these outcomes change lives.
During an internship, I watched seasoned chemists double-check molecular weight as a first step before running any major reaction or setting up a scale-up batch. It doesn’t just affect how much goes into a beaker—it sets the stage for everything downstream.
In academic and industry labs, overlooked molecular weights breed costly accidents or even dangerous mishaps. Some years back, a well-known pharmaceutical lab lost months correcting a production error tied to incorrect molar calculations. These slip-ups rarely stem from carelessness, more often from pressure and long hours. It's easy to brush off the basics, but the fundamentals matter most when stakes run high.
Training up-and-coming chemists on molecular calculations pays huge dividends. Regular refreshers and spot checks foster a safer, more productive environment. Some companies even build in triple-verification protocols. Less glamorous than breakthrough discoveries, but far more valuable in the long run. Digging into the numbers on every label or vial isn’t routine for the sake of routine — it’s habit that provides a backbone for everything that follows.
The molecular weight of 2,3,4-Trihydroxybenzophenone might look like a tiny detail, but it’s the start of real discovery. Chemistry leans on these details, building up from simple numbers to drugs that treat illness or new materials with surprising properties. Getting the basics right makes everything else possible.