Benzil traces its roots back to the golden era of early organic chemistry in the 19th century, paralleling breakthroughs with dyes and solvents. Chemists tracked it through oxidation reactions, using rudimentary techniques by today’s standards, yet grasping the compound’s key features and transformation potential. My own experience digging through historical lab notebooks in grad school reinforced how proud early chemists felt watching color changes signal benzil’s formation during a basic oxidation – a small moment hinting at the broader power within aromatic chemistry. Since then, each decade added detail to our understanding, evolving from academic curiosity to widespread industrial utility, echoing how curiosity drives the field forward piece by piece.
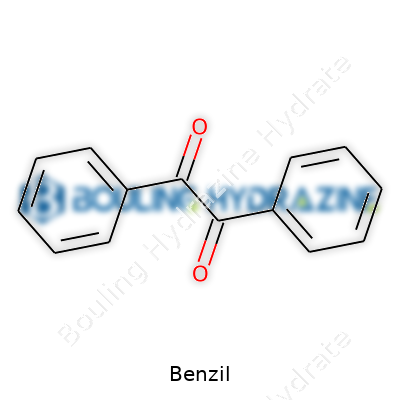
Benzil stands as a yellow crystalline compound, now a staple in labs and factories across the globe. Chemists recognize it as a simple 1,2-diketone with two phenyl rings, which guides both academic research and commercial practice. The substance holds a modest molecular weight and becomes noteworthy not just for its straightforward structure but for its flexible reactivity and clear role as an intermediate. Its role goes beyond the dusty glass vial: benzil acts as a bridge leading to advanced pharmaceuticals, specialty polymers, and photoinitiators that power industries ranging from plastics to electronics.
People often underestimate the importance of physical familiarity with a chemical. Benzil appears as yellow needles or prisms, with a melting point typically a touch above 90°C. Don’t gloss over these numbers; the color and state tell a working chemist a lot about purity and potential contaminants. The scent carries a faintly aromatic note, not easily missed by those who spend enough time in the lab. Its solubility splits the difference between polar and non-polar solvents – it dissolves in acetone, benzene, and ethyl acetate, but sits poorly in water. Two adjacent carbonyl groups drive its core reactivity. The compound shows photoreactivity when hit by ultraviolet light, an angle that has made it a favorite for those working on new polymers or photochemical reactions.
Technical labeling for benzil in research or industry doesn’t just mean slapping a name on a bottle – it’s compliance, traceability, and responsibility. Purity often exceeds 98% for advanced uses, and experienced chemists double-check this with basic melting point tests or chromatography. Any shipment needs clear hazard labeling, as countries adopt variations of the Globally Harmonized System. Documentation often includes batch analysis and spectral fingerprints, which help avoid mistakes and keep experiments – and people – safe. My time in a quality assurance lab drilled home the reality: a simple error in labeling leads to ruined batches or misleading results, and the finer details always matter.
Classic lab routes oxidize benzoin using nitric acid or copper-based reagents, producing benzil through a stepwise set of color changes in solution. Industrial chemistry leans toward scalable, less hazardous methods, using controlled air oxidation or green oxidants where possible. Modern improvements emphasize waste management, and green chemistry shifts encourage switching to less polluting processes. A practical significance hides in these choices: reagent selection, temperature control, and management of byproducts often become the difference between economical manufacturing and logistical nightmares. Over the years, teams chasing higher yields or fewer side products found even modest tweaks could dramatically lower costs and environmental footprint.
Benzil sits as more than just a final product; it’s a pivot point for chemists tinkering with structures. Its diketone core participates in the classic benzil–benzilic acid rearrangement, forming useful alpha-hydroxy acids. That reaction shows up in undergraduate organic texts, but seasoned chemists leverage benzil in producing complex heterocycles, chiral ligands, or photoinitiators. It undergoes nucleophilic addition, reductions, and condensations, transforming into molecules useful for drugs or dyes. A research group I followed took advantage of benzil’s photochemistry, generating reactive intermediates under UV light for specialty plastics. Its role as a starting point – and willingness to follow a chemist’s lead – gives benzil lasting relevance.
Walk into any supply catalog and benzil shows up under various names. You’ll spot ‘diphenylethanedione’, ‘1,2-diphenylethane-1,2-dione’, ‘dibenzoyl’, and of course its common trade names. Recognizing these synonyms helps avoid costly mix-ups in lab procurement or regulatory filings. Marketers sometimes spin catchy names when selling to electronics or polymer sectors, but all point to the same chemical backbone and properties. Learning the full roster of names isn’t trivia – it preempts supply hiccups, dodges regulatory snags, and smooths multinational collaboration.
Work with benzil, and safety matters. The compound doesn’t pose acute dangers like some industrial catalysts, but standard protocols – gloves, goggles, fume hood – make a real difference. Chronic exposure can irritate skin or eyes, and fine dust ought not linger in the air. Workplace monitoring, proper ventilation, and clear hazard signage keep work environments healthy. Teams with robust training history face fewer accidents. On my first warehouse walkthrough, the checklist and signage seemed tedious until you see the alternative: mixed-up labels, absent PPE, and real risks to health. Safety audits, regular reviews, and practical training save time, money, and reputations in the long run.
Benzil’s roles extend far. The manufacturing of photoinitiators takes a considerable slice, powering polymer curing under light for coatings and electronics. In pharmaceuticals, benzil acts both as a building block and sometimes as a chiral auxiliary. The flavor and fragrance sector taps benzil derivatives, while specialty dyes trace their roots to benzil chemistry as well. Analytical labs use it to prepare calibration standards or probe reaction mechanisms. In each field, benzil brings value not as the star, but as an essential enabler connecting raw materials with complex finished goods. My professional circle routinely encounters benzil in synthesis flows: not as an afterthought, but as a planned, optimized step anchoring broader chemical strategy.
New ideas keep benzil relevant decades after its discovery. Research teams explore alternative synthetic routes, less hazardous oxidants, and catalytic systems to lower costs and stress on workers. Scientists studying light-triggered processes return to benzil for its reliable photochemistry and stable radical formation. Advances in asymmetric synthesis look to modify benzil for improved selectivity. Many patents covering new drugs, high-performance plastics, or specialty reagents include benzil as a key intermediate. Each project I contributed to underscored how small changes in this core structure could make or break a large-scale process, and how forward-thinking R&D transforms routine chemistry into next-generation applications.
Benzil slips beneath the public radar for toxicity, yet responsible companies and researchers pay close attention. Acute exposures tend not to provoke drastic effects, but chronic contact deserves respect—dermatitis, mild mucous membrane irritation, or, in unlucky cases, sensitization. Animal studies show relatively low systemic toxicity, but I learned early on not to grow complacent with organic chemicals. Disposing of benzil waste, managing spill risks, and monitoring occupational exposure rates follow both legal mandates and the ethics of working in science. Up-to-date safety data help companies stay in compliance, while open communication between workers, management, and regulatory bodies ensures research keeps people in mind, not just molecular targets.
Benzil’s future rides on adaptability. As industries shift toward greener practices, the way benzil gets produced, handled, and deployed will change as well. Ongoing work to design recyclable or safer reagents influences how companies commit to new processes. Advances in photoinitiator technology for 3D printing and flexible electronics look to benzil’s core – chemists now pursue modifications fine-tuned for energy efficiency or environmental compatibility. Pharmas shed outdated methods in favor of safer, more atom-economic benzil transformations. Conversations I join at professional conferences reveal a consensus: despite its long history, benzil belongs on the cutting edge for decades more if researchers and companies keep investing in fresh ideas and higher standards.
Most people walk past science supply stores without giving a second thought to ingredients with names like benzil. I first came across benzil in my undergraduate chemistry lab. We were synthesizing dyes, and my instructor handed out a pale yellow powder I’d never seen. Turns out, benzil is a backbone for many reactions we all rely on in ways we might never notice.
Benzil’s real value lies in its role as a starting block in organic chemistry. Fact: it works like a hinge that swings open dozens of doors in a lab. Chemists call it a diketone—basically, two connected carbonyl groups—making it reactive but manageable. Some of the biggest uses show up in the production of pharmaceuticals, dyes, and even special polymers. Any student who has performed the well-known benzilic acid rearrangement gets a taste of how a structure like benzil can transform into complex molecules that show up in pain relievers and anticoagulants.
In industry settings, benzil finds a purpose in the manufacture of photoinitiators. These are compounds that help kickstart chemical reactions under light. Speak to any technician creating certain plastics or resins and you’ll hear about benzil-based solutions for things like dental restorations and adhesives. The high stability and ease of handling make benzil appealing to companies scaling up production. There's a good reason why modern electronics and coatings use compounds born from benzil chemistry.
I’ve seen plenty of students get careless with chemicals, thinking the hazards are overblown. Benzil’s low toxicity relative to other industrial chemicals might reinforce that attitude, but experience shows caution still belongs. In large-scale manufacturing, oversight matters: run-off, improper disposal, and emissions can add up over time. Researchers have flagged some metabolites of benzil as cause for concern in aquatic systems. From what I've seen, simple practices—proper waste capture, using minimal effective quantities—make a measurable difference.
Transparency in sourcing also needs attention. Some benzil production routes still start with petroleum-based feedstocks, making the carbon footprint bigger than it needs to be. Nowadays, researchers and manufacturers actively look for greener synthesis pathways—using renewable feedstocks or optimizing processes to cut energy use and byproducts. Regulatory guidelines in many countries now reflect tighter standards on sourcing and emission controls.
If more people thought about the chemicals behind their daily products, demand for sustainable options would likely increase. The chemistry community has started spotlighting how small changes at the start of a supply chain, like greener benzil manufacturing, ripple out into safer products and less environmental fallout. Universities and trade groups offer training on handling and disposal, but most improvements start on the shop floor. Sharing best practices, investing in updated equipment, and pressing for accountability in material sourcing can deliver real-world results.
Benzil may seem like an obscure chemical, but its footprint runs through industries that shape modern life. Taking a little extra responsibility at every stage—design, production, and waste handling—can help turn an old staple into a benchmark for safer, smarter chemistry.
Benzil, with the formula C14H10O2, first grabbed my attention during a college lab when a yellow crystal formed in my beaker after a few hours of careful mixing and heating. Today, benzil’s striking yellow color and its structure—a pair of benzene rings joined by two carbonyl groups—makes it more than just another line in a chemistry book. Those memorable double benzene rings give benzil its solid place in synthetic organic chemistry.
C14H10O2 packs a punch. This formula represents a stepping stone for a wide range of chemical reactions. During my years in research, benzil often worked like a Swiss Army knife. I remember using it as a model compound to help new students understand reactions like nucleophilic addition, which turns these yellow crystals into all sorts of new molecules. Because it contains two reactive carbonyl groups, it stands out as ideal for teaching fundamental chemistry concepts and exploring more advanced organic synthesis.
You’ll find benzil used both as a teaching tool in countless organic chemistry courses and as a key player in real-world production. It’s not just a theoretical exercise; benzil plays a role in manufacturing dyes, pharmaceuticals, and plastic additives. Aside from the teaching labs, its photo-initiating properties drive polymer science and industrial photochemistry. Over the years, I’ve watched startups and research groups turn benzil into the backbone of experimental protocols for new polymers and light-activated materials.
As much as I admired benzil in the lab, I always wondered about its environmental footprint. Like many aromatic compounds, its production relies on benzene and other substances tied to the petrochemical industry. The push toward greener chemistry calls for better, more sustainable practices, and benzil sits squarely in the middle of that conversation. Advances in solvent recycling and catalyst optimization show promise, but most labs still use traditional routes for benzil synthesis. Years of handling it also made me aware of personal risks: gloves and goggles became second nature once I read up on the safety sheets. Chronic exposure calls for careful protocols because aromatic compounds can lead to long-term health hazards.
Science always moves forward by learning from experience and pushing boundaries. I’ve seen creative chemists switch to greener solvents and test new catalysts, trying to ease benzil’s environmental impact by slowly replacing petroleum-based starting materials. It’s no easy task, since large-scale change takes collaboration and funding, but the push for sustainability grows stronger every year. If schools and industrial plants push for eco-friendlier methods, benzil will keep its value in classrooms and production lines, just without the heavy cost to health or nature.
Benzil and its formula C14H10O2 serve as a reminder that chemistry is about more than equations on a chalkboard. It’s about smart choices, hands-on learning, and keeping an eye on how science shapes the bigger world outside our labs. The future of benzil lies in the hands of those willing to push for safer, cleaner, and smarter chemistry.
Benzil always caught my eye in the chemistry lab — bright yellow crystals, smooth as silk under the microscope. Its reputation for helping with organic syntheses made it a staple in teaching labs and research benches. But handling benzil goes beyond marveling at its looks or watching reactions unfold. Safety deserves more than a passing thought, especially for those new to the game.
Early days in my chemistry studies, my mentor used to remind the class, “Gloves and goggles aren’t optional here, folks. They’re as important as the chemicals themselves.” He had a point; lab safety never follows a ‘better safe than sorry’ approach. Safety starts from the ground up, building on respect for every chemical in the cabinet. Benzil doesn't break this rule. Its low toxicity rating often puts people at ease, but that’s where people slip up. Benzil dust feels like harmless gold powder, but it can cause mild skin irritation for some. Inhaling too much powder, especially during weighing or transfer, may trigger headaches or cause mild irritation in the nose and throat.
The main risk lies less in the acute effects and more in the lazy habits that develop over time. I’ve watched colleagues forgo eye protection, letting a tiny crystal bounce right toward the face when scraping it from a watch glass. The sense of comfort from repeated use can be dangerous. Years in the lab taught me that even “mild irritants” escalate if handled carelessly day after day. A single spill seems trivial until it happens without gloves, and then you get to know irritation firsthand.
Science pushes us to dig into the literature: animal studies, workplace reports, and occupational exposure guidelines. From what’s published, benzil ends up in the middle of the risk scale. Not the worst, not the safest either. Most regulatory agencies don’t list it as cancer-causing or as a compound with extreme environmental concerns. Still, nobody recommends eating it, rubbing it on your hands, or ignoring accidental splashes.
Experience in the lab teaches that most chemical risks add up from small incidents: dust when weighing, residue left behind on the bench, contaminated gloves touching a water bottle. Proper lab hygiene — gloves, goggles, working under a fume hood — keeps these chances low.
One improvement I’ve seen over the years: better labeling and clear protocols for every experiment, no matter how routine. Supervisors set the tone. They create spaces where people aren’t embarrassed to double-check, or to ask for newer gloves. Running safety drills and practicing clean techniques with even “low-risk” materials helps prevent the “harmless chemical” mindset.
Lab culture shifts when people treat every compound — benzil included — with respect. Supervisors and peers encouraging regular handwashing, using disposable spatulas, and keeping food out of the lab can break the chain of accidental exposure that sneaks up in unexpected ways.
Old habits aren’t that hard to change if you know the risks. Approach benzil and similar chemicals as you would any tool: with attention and respect for the possible downsides. Gloves and goggles don't just protect against disasters; they stop the small events from building up over years. Keep the processes simple, share what you know, and maybe the next group of students or researchers won’t learn about benzil’s drawbacks the hard way. Lab safety always has room to grow, but it starts with everyone deciding the rules matter even for the chemicals that seem friendly on paper.
Benzil comes out of a classic lab trick that sits close to any chemist’s heart. You start with benzoin, a material that shows up in many basic organic experiments. The transformation from benzoin to benzil is all about a clean oxidation step. I’ve seen this reaction run on dingy lab benches and in high-tech facilities, and the real difference boils down to the details—how clean you want your product and how tight your budget feels.
There’s more going on here than just “making a product.” Benzil turns into a stepping stone for building pharmaceuticals, dyes, and even tools for exploring molecular machines. Students running their first oxidation feel the stress when purple fumes drift up, and everyone knows mistakes in this reaction can fill a room with a sharp, almond smell. You’re not just mixing chemicals; you’re setting up the foundation for dozens of research paths. That matters because better methods start with basics like these. Get the basics right, and suddenly a whole chemical family opens up.
The heart of the process? An oxidizing agent like nitric acid gets poured over benzoin, usually with steady stirring and gentle heat. Nitric acid works quickly but punishes anyone who skips safety goggles. More modern recipes swap in copper(II) sulfate with sodium bromide and a splash of alcohol. This milder version feels friendlier and skips some of the harsh fumes, which makes it attractive for teaching labs or DIY projects. Old-timers prefer the classic route for its reliability. Both ways rely on taking benzoin and stripping away hydrogens to make those valuable twin carbonyl groups sit side-by-side.
Nobody wants to see nitric acid splashing around where it shouldn’t. Nitrates run into rivers and build headaches for water treatment folks. Those clean green labels we see today push for gentler oxidants, fewer fumes, and reactions that run without wasting water or heat. I remember conversations with green chemists who push for hydrogen peroxide as an oxidant, or using electric current to make the transformation. These tweaks aim to cut waste, save energy, and dodge nasty byproducts. Up-and-coming research in photochemistry even uses light to drive the oxidation, dodging hazardous reactants altogether.
Lab instructors care about yield and purity because a clean benzil sample can mean the difference between successful research and dud results. Commercial producers want reaction cycles they can repeat without costly hiccups. The way you make benzil changes just how much work you do before your next big experiment. Impurities trip up sensitive reactions, muck up pharmaceutical syntheses, and skew lab results. This forces research teams and students alike to double-check every step, clean glassware obsessively, and weigh out chemicals with sharp-eyed attention.
There’s no single “best” way to make benzil, but new technology points toward greener, safer, and cheaper methods. Shared experience among chemists tells us that careful planning, attention to detail, and investment in safer chemicals pays off. As regulations tighten on hazardous waste, research teams now hunt down oxidations that work in water or use electricity. Every time the recipe gets safer or more sustainable, it sets a better standard for the next generation of chemists.
Ask folks in manufacturing or chemistry about go-to ingredients, and benzil almost always finds a mention. Its yellow crystals might seem unremarkable, but behind the scenes, they drive reactions that keep whole industries running. My first real exposure to benzil came in a university lab, watching it act as the star of a photoinitiator experiment—one that set the groundwork for fields like plastics and 3D printing. This compound isn’t as headline-grabbing as fancy new materials, but few can match its versatility.
Modern manufacturing banks on plastics with precise properties, from soft medical components to rigid automotive parts. Benzil stands out for helping get there. It works as a photoinitiator, where light triggers chemical reactions—think of the quick setting of dental fillings or the neat layers in 3D-printed models. By skipping the need for heat or long waits, benzil saves both time and energy.
This ability matters outside the lab, too. In printing inks, benzil lets printers cure colors under UV lamps almost instantly. That boost in speed can mean lower costs and less wasted energy for companies, offering a rare mix of efficiency and reliability. Big names in the printing industry rely on it to keep their processes running at scale.
Pharmaceutical research might sound worlds away from plastics, but benzil helps shape both. It’s a key ingredient in organic synthesis, helping construct complicated drug molecules. Take anticonvulsants or certain sedatives—chemists use benzil as a building block to reach molecules the body can use. Researchers value benzil’s stability and predictability, reducing surprises during manufacturing.
In my own grad school experience, the difference between a clean, high-yield benzil reaction and a messy substitute was the difference between a passable result and a breakthrough. With cost pressures rising and requirements for purity tightening, benzil continues to earn its place in labs worldwide.
Benzil’s usefulness comes with responsibilities. To see its impact from another angle, it helps to consider the waste produced in big plants. Benzil itself isn’t the most toxic reagent, but every chemical steps into a larger system—spills, improper disposal, or poor ventilation can trigger real problems. Feedback from industry safety audits often points to a need for stricter controls and better worker education.
Sustainability also enters the debate. Making benzil typically starts with petroleum-derived compounds, raising questions about long-term environmental costs. Creative solutions surface from time to time—ongoing work in green chemistry aims to tweak the process, cutting hazardous byproducts and using renewable feedstocks. Pilot projects in chemical plants already show that these greener routes can compete with traditional methods, though scaling up remains a work in progress.
Industry veterans know that rare is the compound as humble as benzil yet as crucial across so many sectors. It keeps printers working, drugs flowing, and manufacturers meeting deadlines. As factories chase lower carbon footprints and tighter safety standards, the real challenge becomes balancing performance with responsibility. Supporting innovation in benzil production—especially those that avoid heavy metals or fossil fuels—means investing in safer jobs, cleaner waterways, and a better shot at sustainable growth.
Sometimes, progress relies less on eye-catching new inventions and more on getting the most out of proven workhorses. Benzil is a solid example of how industry can do both.