Ethylene sulfate hasn’t always sat in the spotlight. Early texts in organosulfur chemistry often skipped right past it, giving more attention to simpler sulfates or larger, more familiar molecules. Back in the 1940s and 1950s, industrial researchers noticed the unique reactivity of cyclic sulfates, including ethylene sulfate, though large-scale synthesis at the time hit several snags. The process for making ethylene sulfate depended heavily on finding reliable ways to handle the raw materials safely, especially as manufacturing environments became more safety conscious. Through the years, improvements in purification and analytical techniques, like NMR and advanced chromatography, helped chemists get a closer look at the compound’s structure and behavior. Eventually, streamlined preparation methods and tighter controls meant folks could use ethylene sulfate in both research and commercial applications without as much worry about dangerous byproducts or unpredictable impurities.
As a small, sulfur-containing cyclic ester, ethylene sulfate stands out mostly for its high reactivity and compact size. You’ll spot it as a colorless crystalline solid or sometimes as a transparent liquid, depending on how pure and how warm you keep it. If you've worked in a lab with battery chemicals or specialty organics, you may have come across a bottle of this stuff somewhere on the shelf. Its main attraction comes from the strained three-membered ring and the electrophilic character the sulfur atom brings to the equation. These properties make it interesting for chemists hunting for routes to more complex compounds and for people trying to improve electrolyte formulations in energy storage systems.
Ethylene sulfate packs a punch in a small structure. Its melting point usually falls in the moderate range for organics, rarely straying into territory that causes headaches with storage or transport. It holds a decent solubility in polar solvents, letting it mix well in the sorts of reaction setups that can otherwise slow researchers down. In terms of reactivity, the molecule flips between being a helpful synthetic intermediate and a source of trouble if overlooked on the bench. I remember one afternoon when spilled ethylene sulfate met a bit of water on a worktable—watching the decomposed mess eat through careless paperwork made an impression. Handling it demands attention to cleanup routines and thoughtful containment rather than the “set it and forget it” approach sometimes seen with more stable esters.
Labels on bottles of ethylene sulfate carry real warnings for good reason. The regulatory standards, at least in labs I’ve worked in, ask for clear hazard symbols and detailed handling instructions right next to the lot number. Chemists can’t look at a bottle and treat it like vegan-friendly soap. Accurate labeling includes warnings about possible respiratory irritation, eye harm, and skin contact issues. For facilities following strict compliance rules, safety data sheets get checked along with each new order. Any mistake in storage—like letting it sit too close to moisture or heat—risks damaging not just the product but also nearby equipment. These careful steps might sound like overkill until you’ve seen what a surprise reaction can do.
Getting pure ethylene sulfate involves a careful sequence of steps. Most routes start from dihydroxyethylene or a related compound, using strong sulfurylating agents to foster ring closure. Managing heat and timing in these reactions spells the difference between a successful batch and an unusable slurry. In industrial-scale settings, engineers introduced continuous flow systems to better control exothermic surges. Sometimes the work comes down to patience and willingness to tweak pH, concentration, and reaction vessels. On the smaller scale, glassware selection and agitation techniques matter just as much as choice of reagents. I found out early how much difference a slightly corroded stirrer can make to yields and purity—details too easy to ignore until purification days get unexpectedly long.
Ethylene sulfate refuses to stay put. Electrophilic sulfur gives it a key role in transformations like nucleophilic ring openings, often using water, alcohols, or amines as attacking reagents. This lets it act as a gateway to diols, hydroxyalkyl sulfonates, or other value-added groups. In electrochemical settings, the compound provides room for ion-conducting moieties, supporting performance in battery prototypes. Skilled chemists sometimes reach for ethylene sulfate to introduce a controlled amount of sulfur into scaffolds for further diversification. From my own experiments, I see room for frustration, too—side reactions easily pop up, especially if care slips during stoichiometry calculations, and unplanned path ways can complicate purification. Yet these characteristics make the compound useful for method development, especially in efforts to build libraries of new organosulfur compounds.
Ethylene sulfate goes by more than one label. Industry slang sometimes calls it “1,3,2-dioxathiane 2,2-dioxide,” matching older literature. Some catalogues list it simply as “ethylene cyclic sulfate.” Concrete, reliable chemical structure information is what separates legitimate supply from uncertain knockoffs; using IUPAC standards for names and formulas prevents mix-ups between close but distinctly different chemicals. Anyone filing paperwork with safety officers or preparing compliance records benefits from double-checking these naming rules.
Ethylene sulfate demands respect. Its stability wobbles around moisture and high temperatures, pushing lab managers to drill careful storage and swift spill cleanup habits into new hires. Standard procedures I’ve seen include wearing gloves, using goggles, and keeping good ventilation any time the material lands on a benchtop. If spills get missed, corrosion follows soon after—labels on surface-sensitive tools sometimes fade faster if the compound lingers in the same spot. Safety audits make a point of checking separation from incompatible chemicals like strong bases and reactive metals. In training sessions, we talk less about generic hazards and more about memories of burned gloves, scarred bench surfaces, and near-miss inhalation episodes. A culture of sharing close calls and lessons learned protects more than any rules printed on a label.
People rarely buy ethylene sulfate as a final destination; it acts as a springboard in both research and industrial settings. In lithium-ion battery labs, tweaks to electrolyte blends often rely on small doses of this compound to push conductivity higher and extend shelf life. Polymer chemists turn to it for tuning block copolymer architectures in a push to discover next-generation functional materials. In pharmaceutical research, the molecule shows up as a building block for more elaborate molecules where a sulfur moiety brings desired activity or solubility. Each application means weighing the reactivity and hazard against the benefits. In my own group, we saw successful breakthroughs in cross-linking experiments for specialty coatings, thanks to ethylene sulfate’s lively nature—though not without a storeroom filled with failed attempts and plenty of near-miss safety talks.
Interest in ethylene sulfate waxes and wanes with trends in energy, materials, and green chemistry. Over the past decade, the surge in battery research rocketed demand for unusual electrolyte components, and that highlighted this compound's unique features. Scientists dug into modification techniques, hoping to fine-tune stability, minimize environmental hazards, and unlock more durable battery lifetimes. Researchers published studies on new ring-opening strategies, solvent-free synthesis routes, and greener sulfonylation methods. These advances trickle from specialty journals into mainstream textbooks and eventually into the pilot plants I’ve visited. Collaborations between academic theorists and real-world engineers keep the field moving, testing out ideas on how to wrangle ethylene sulfate into friendlier, more sustainable forms.
For all its usefulness, ethylene sulfate poses genuine health risks. Published studies report that it sits in the moderate-to-high hazard range among cyclic sulfates, with risks concentrated mainly on direct exposure to skin, eyes, or respiratory tracts. Animal study trends and cell culture assays signal some potential for harm from chronic low-level exposure—an uncomfortable prospect for shift workers and lab folks alike. Researchers have examined breakdown products under simulated environmental conditions, flagging several as acutely toxic. Memory serves of times colleagues repeated exposure scenarios with better PPE only after uncomfortable symptoms cropped up. Proper disposal and treatment protocols keep these risks somewhat in check, but urgent calls for improved, less hazardous alternatives remain.
Many eyes track the possible future of ethylene sulfate, and for good reason. Emerging battery designs and polymer materials depend on specialty chemicals that combine high performance with improved safety profiles. There’s movement toward methods that lower the environmental cost of making and using ethylene sulfate, including solventless reactions and renewable feedstocks. I have seen start-ups and large firms alike fund collaborative efforts to engineer derivatives with reduced toxicity, hoping to capture both market share and a cleaner conscience. The compound looks set to remain a mainstay in sectors hungry for ever-more clever intermediates—if researchers and producers stay disciplined about minimizing the hazards and meeting tighter standards for environmental stewardship.
Ethylene sulfate doesn’t get called out in headlines, but in my years reading up on battery science and talking to folks in the energy sector, it keeps popping up behind the scenes. The world keeps spinning faster on lithium-ion batteries—phones, laptops, electric vehicles—and this chemical is part of the underpinnings. Ethylene sulfate threads its way into the conversation because it helps batteries run safer, last longer, and deal with tougher conditions.
Ask any chemist working in batteries about challenges, and they’ll probably mention that balancing energy density with safety and shelf life turns out tricky. Ethylene sulfate serves a specific role as an electrolyte additive. Inside lithium-ion batteries, the electrolyte ferries lithium ions from one side to the other—it’s a highway for the things that keep our gadgets running. Additives shape how that highway holds up over time. Scientists found that a pinch of ethylene sulfate encourages the formation of a better protective layer on the battery’s anode—the “solid electrolyte interphase.” That’s technical, but the point is, the layer keeps the battery from breaking down as quickly or catching fire if things heat up.
This stuff isn’t just insurance for your phone. Carmakers and grid technology companies are paying more attention because everyone hates the thought of battery fires or quick battery death in electric vehicles. Every extra year that a car battery survives means less mining for scarce metals and less electronic waste piling up.
Here’s where it gets real: with the push toward electrifying cars and renewable energy, battery factories keep popping up. Every efficiency gain means more affordable technology for everyday people and less pollution. If ethylene sulfate can help squeeze out better results—maybe by cutting the risk of batteries swelling, leaking, or shorting out—manufacturers have fewer headaches and customers end up with safer products.
Of course, there’s responsibility attached. Using any chemical at scale means keeping an eye on the supply chain and safety. In the United States and Europe, you don’t get away with reckless use in consumer goods, which keeps pressure on companies to test and monitor. Researchers look into alternatives and improvements all the time, so the use of ethylene sulfate stays under review.
No single compound will solve the world's energy storage needs, but small changes can ripple out and make a difference. The lesson I keep learning is that big change in technology usually hides in careful choices, unassuming molecules, and constant tinkering. Ethylene sulfate is hardly a household name, but as more of us switch to electric cars or rely on our devices for daily life, the work of chemicals like this adds up.
The future of reliable and safe batteries depends on constant improvement. Supporting research, encouraging safety, and demanding clear labeling all help empower people and communities who rely on each new gadget. Ethylene sulfate proves that even the ingredients no one talks about can help write the next chapter of clean energy and tech.
Most people are more familiar with chemicals like bleach or gasoline than names such as ethylene sulfate. Still, this compound finds its way into scientific labs and some corners of battery research, and questions around its safety keep popping up. Even if you don’t work with specialty chemicals or get involved with manufacturing, chemical risks have ripple effects that touch everyone through workplace standards, environmental rules, and consumer protection. If a substance poses hazards, important choices follow about where and how it can be used.
Ethylene sulfate isn’t a household name like lead paint or asbestos, but its health risks deserve attention, especially for anyone handling or transporting it in an industrial setting. Scientific literature and regulatory agencies point to clear reasons for caution. Ethylene sulfate acts as an irritant. Skin, eyes, and the respiratory system can all suffer from direct exposure. Some animal tests indicate corrosive or damaging effects. Accidental contact can burn skin, damage membranes, and trigger severe irritation. Breathing in the vapors may cause coughing and breathing difficulties.
As for longer-term or indirect risks such as cancer or chronic harm, research remains scarce and inconclusive. No one has pinned down every consequence — but waiting for definitive answers before taking precautions doesn’t make sense. Regulatory bodies rely on available data to err on the side of caution. For example, the Globally Harmonized System for chemical classification cites ethylene sulfate as hazardous, with recommendations to use gloves, goggles, and proper ventilation and to keep it away from environments where it might react dangerously.
Early in my career, I watched a spill of a lesser-known solvent trigger a scramble — folks suddenly wanted to know what it did to groundwater or whether fumes would reach offices nearby. Ethylene sulfate belongs in the same camp. Its reactivity and solubility raise flags about what could happen if a leak reached soil or water. Though it’s not as notorious as pesticides that persist for decades, the risk of localized, acute damage matters just as much for workers and nearby residents.
Regulatory bodies are starting to catch up, but not always at the speed that frontline workers or emergency responders need. One major challenge lies in inconsistent labeling or missing information in global transportation networks. Laws in one country may call for warnings, while another jurisdiction lets shipments move with sparse information. That patchwork leaves too many people guessing about safety steps.
Worker protection groups push for greater transparency. Clearly marked hazard labels, standardized training for anyone moving or using ethylene sulfate, and easy-to-access emergency handling procedures would help reduce risks. Companies and labs benefit by avoiding accidents and lawsuits down the road. Local governments can set standards that go above mere compliance — regular drills, controlled storage, and quick reporting of spills build confidence and keep small mishaps from growing into emergencies.
New chemicals and materials seem to show up every year. Companies may chase breakthroughs, but safety basics never go out of style. Wearing basic protective gear, understanding what chemicals can and can’t mix, and getting fresh air circulating remain core practices. Simple investments in safety training and equipment pay off over time. Those steps protect not only individual workers but also the communities near manufacturing or research sites.
The science on ethylene sulfate continues to develop, but its hazardous reputation already stands. Respecting its risks means staying informed, pressing for clear rules, and building a culture of safety from the ground up.
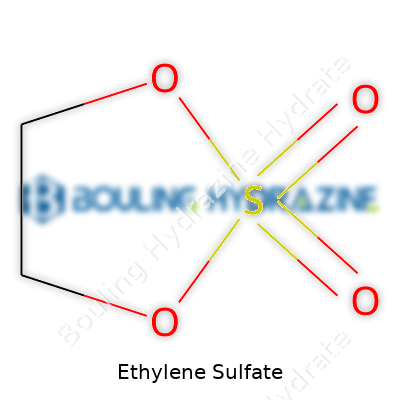
Ethylene sulfate might not pop up in everyday conversation, but in the world of chemistry and battery research, it carves out a small yet meaningful spot. The chemical formula for ethylene sulfate is C2H4SO4. That shorthand packs more significance than most realize. Getting this formula right doesn’t just matter for exams or trivia—it actually impacts research progress, product safety, and our understanding of new technologies.
Back in college, I’d spend hours memorizing chemical formulas for organic and inorganic compounds. I hated the rote memorization, but as I joined a research lab, it dawned on me that the formula isn’t just a name. C2H4SO4 spells out the molecule’s backbone. Two carbon atoms, four hydrogens, one sulfur, four oxygens. This arrangement matters because ethylene sulfate sits at a crossroads between sulfur chemistry and ethylene derivatives. The small tweak of adding or removing an atom can flip a compound from safe to reactive—or from useful to hazardous.
Readers might ask: Why care about ethylene sulfate at all? Turns out, it pops up in the manufacturing of electrolytes for lithium-ion batteries and as a reagent in organic synthesis. In batteries, researchers test compounds to improve stability, safety, and longevity. Mistakes in chemical notation or misunderstanding the exact composition lead to failed experiments, wasted years, or worse—dangerous products. Battery fires from incorrect formulation are no joke; labs take extra care to make sure each compound aligns exactly with what the formula promises.
One false step—swapping a number, adding a stray atom—and the result can invalidate months of work. I’ve seen journal retract papers because the chemistry didn’t check out under scrutiny. Mistakes ripple out, sometimes forcing regulatory agencies to step in. According to studies published in peer-reviewed journals, errors in chemical formulas slow down the pace of research and waste millions in funding every year. The domino effect stretches far beyond the lab: think about the risk for health, security, and technology sectors.
Students, researchers, and professionals all benefit from a solid grasp of the formulas they use. Cross-checking information, especially from top databases and open-source repositories, goes a long way to prevent slips. Teachers can create more immersive labs so students move beyond memorization and actually connect structural formulas to real-world applications. Universities and industry labs should invest in better peer review, data verification, and collaboration between chemists and engineers, which helps cut down formula errors and supports safer product development.
Precision in language defines science. Calling out the formula of ethylene sulfate as C2H4SO4 sounds small, but it forms the foundation not just of one experiment, but of scientific progress itself. Instead of glossing over “just another” chemical name, every student and researcher should appreciate that accuracy here sets up everything else. In a field where atoms make all the difference, the details become the story.
If you’ve spent any stretch of time working in a lab, a chemical plant, or even a modest battery R&D room, you know ethylene sulfate isn’t something you just leave lying around. In the right hands, this chemical does a serious job, especially in the battery sector. It’s no secret that it can boost the stability of lithium batteries, but that usefulness means nothing if you mishandle it and pay the price in burned skin or worse. I’ve seen more than one rookie assume a simple pair of gloves and a grin is enough—trust me, it’s never that easy.
Ethylene sulfate doesn’t play nicely with water. You add moisture, even accidentally, and you’ll set the stage for decomposition or some chemistry you definitely didn’t plan for. I've worked with folks who kept it too close to a bathroom or left containers uncapped after hours—by Monday morning, the residue had corroded through metal shelving. So, cool and dry is the name of the game. Stick it in a tightly sealed container, away from damp rooms, leaky pipes, or wide-open windows. This isn’t about being overly cautious; it’s about learning from mistakes you don’t want to repeat.
Using a random bottle or leftover beaker from another experiment equals disaster. Ethylene sulfate reacts with some plastics and uncoated metals. Glass containers, with the right seals, hold up best from what I’ve seen. Always label things the moment you pour it. I once dealt with a colleague who left an unmarked, sticky flask in the fridge; that oversight cost us half a day of cleanup and a lengthy safety review. Labels aren’t decoration—they’re there so no one has to play guessing games with safety.
Contact with ethylene sulfate brings pain you won’t forget. It’s tempting to ditch the glasses or skip a face shield during short tasks, but I’ve seen what one quick splash can do. Disposable nitrile gloves, thick lab coats, and full-seal goggles are non-negotiable. In small research labs, where supervisors aren’t always hovering, it falls to coworkers to keep each other honest. Goggle up and put away the bravado. Your eyes will thank you.
Too many labs believe a ceiling fan counts as ventilation. Real extraction hoods, properly maintained, give you space to handle spills or fumes. Ethylene sulfate vapors do more than stink up the place. They can cause headaches or hit your lungs even if you don’t notice at first. A well-ventilated work zone, with airflow tested every month, keeps accidents from becoming emergencies.
Regulations keep changing, and ethylene sulfate is on plenty of watchlists around the world. Before bringing it in-house, check for the latest updates from occupational safety agencies. Don’t trust outdated PDFs or word-of-mouth rules. In my own experience, local officials sometimes spot-check for updated protocols, and missing paperwork can shut down your operation overnight.
It’s easy to get complacent, especially in teams who think they’ve seen it all. Regular drills and refresher sessions make a real difference. Incorporate hands-on spill response and decontamination routines. Set honest, direct consequences for shortcuts instead of relying on scare tactics or vague warnings. The more open you keep these conversations, the fewer injuries or fines you’ll weather year to year.
Ethylene sulfate isn’t a chemical you find in the cleaning aisle or hear about in everyday conversations. In my time around labs and reading countless technical journals, it always stuck out for being both handy and, frankly, a bit underappreciated. This compound comes to life as a colorless liquid, reminding you a little of dense syrup but thinner in texture. Pick up a vial in a controlled environment, and you’ll notice it doesn’t give off that overpowering odor found in many sulfates, which makes handling less unpleasant.
Chemists care about boiling and melting points – it’s the difference between a smooth day in the lab and a surprise mess on the bench. Ethylene sulfate boils at lower temperatures than many other cyclic sulfates, tipping over into vapor mode much quicker than you’d prefer if you’re not watching that hotplate closely. Its melting point sits lower than ice, so you’ll rarely see it as a solid unless you lock the lab in a freezer. That leaves you with a chemical that stays liquid unless you start pushing the thermostat way down.
When anyone mentions solubility, don’t tune out. I’ve watched enough colleagues wrestle with stubborn compounds to know it’s a headache when something refuses to mix. Ethylene sulfate, though, plays nice with a lot of liquids. Toss it in water, and it dissolves fairly well, which matters if you’re working on battery electrolytes or specialized reagents. Mix it with organic solvents, and the hassle drops even more. Its molecular structure makes it more cooperative than you might first suspect, letting you blend it into diverse environments without endless tinkering.
There’s another side. Once you start handling chemicals that move between states with only a gentle nudge, like ethylene sulfate, lab safety isn’t a throwaway line. Even though it appears pretty stable, its vapors can sneak up on you if you heat it without proper ventilation. I can recall a day a colleague kicked off a little too much vapor and learned the hard way why fume hoods aren’t just for show. Its density, heavier than water, means a spill doesn’t simply vanish – it creeps down drains and pools in places you don’t want it. Clean-up demands real attention.
This compound attracts interest because battery makers want stable, versatile components, and ethylene sulfate offers that through its liquid nature, manageable viscosity, and good solubility. At the same time, its tendency to react strongly with certain bases can catch technologists off guard. There’s no shortcut: robust training and respect for its fast phase-changes lower the risk of lab mishaps. Labs working with this chemical benefit from adopting air monitoring tools and routine refresher drills on chemical hygiene – a practice I’ve watched save both experiments and people from nasty surprises.
Ethylene sulfate, for all its quirks, opens up fresh paths in chemical engineering and energy storage. Every property – from its volatility to its cooperative mixing behavior – shapes how researchers build safer workplaces and more efficient processes. Understanding its strengths and hazards stays vital, not just for progress but for keeping a promise to everyone in the lab: no science project is worth risking health or safety.